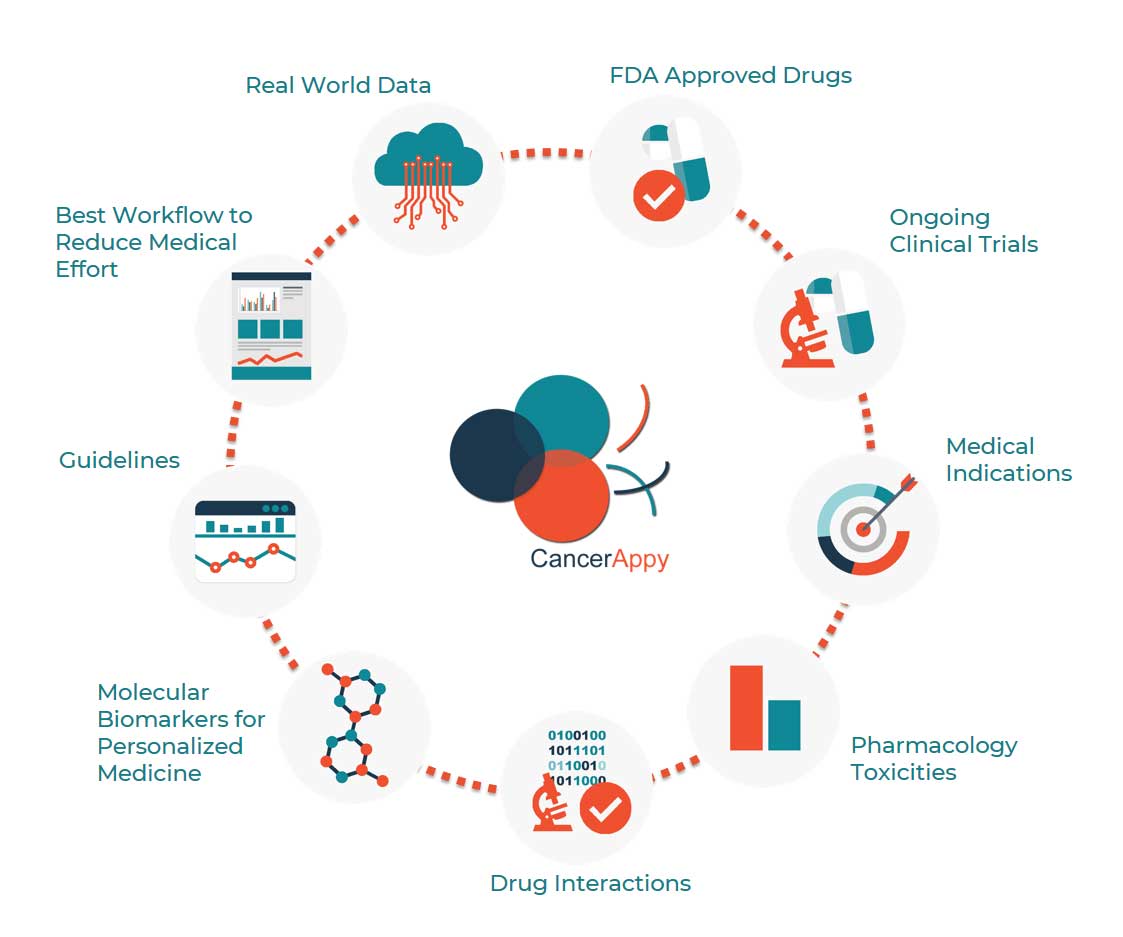

Integrated and processed information on the following topics
- FDA Approved Drugs
- Medical indications
- Pharmacology toxicities
- Drugs interactions
- Ongoing clinical studies
- European and American guidelines
- Molecular Biomarkers for personalized medicine
- Real world data
- Best workflow to reduce medical effort
- Genomic Molecular Alterations
- Evaluation of oncogenic molecular panels
- Druggable vulnerabilities
- Targeted agents
- Therapeutic opportunities
- Identification of best treatment based on proprietary algorithms
- Potential target inhibition
- Current clinical development
- Competitive landscape
- Target population and indication
- Evaluation of attrition rate
- Pharma intelligent
- Intellectual property protection•
- Licensing and business deals
- Identification of best clinical development
.